By Christina Towers, PhD.
The degradation of cellular proteins is a critical step of both regulation and quality control and results in the turn over and recycling of critical amino acids. The two main mechanisms of protein degradation converge on either the proteasome or the lysosome, the latter of which can be further subdivided into macroautophagy, microautophagy, and chaperone mediated autophagy.
Macroautophagy is the process by which double membrane vesicles called autophagosomes deliver damaged proteins and organelles to the lysosome for degradation. Microautophagy also delivers material to the lysosome via vesicles, but in a slightly more direct manner, using vesicles formed from invagination of the lysosomal membrane itself. Distinct from the first two forms of autophagy, chaperone mediated autophagy (CMA) does not use vesicles, but instead uses chaperone proteins to directly target specific proteins to the lumen of the lysosome. One of the most important distinctions is that CMA targets very specific proteins for degradation, only those with a conserved penta-peptide motif in their amino acid sequence.
The process of CMA begins with the heat shock-cognate protein of 70 kDa (Hsc70) binding to the consensus motif to act as a chaperone that guides the targeted proteins to the lysosome-associated membrane protein type 2A (LAMP-2A). Once docked on the outside of the lysosomal membrane, the targeted protein begins to unfold before it is internalized into the lysosomal lumen with the help of other chaperon and co-chaperone proteins, including Hsc90. Proteases and hydrolases that function at low pH in the lysosome then degrade these proteins, recycling critical amino acids1.
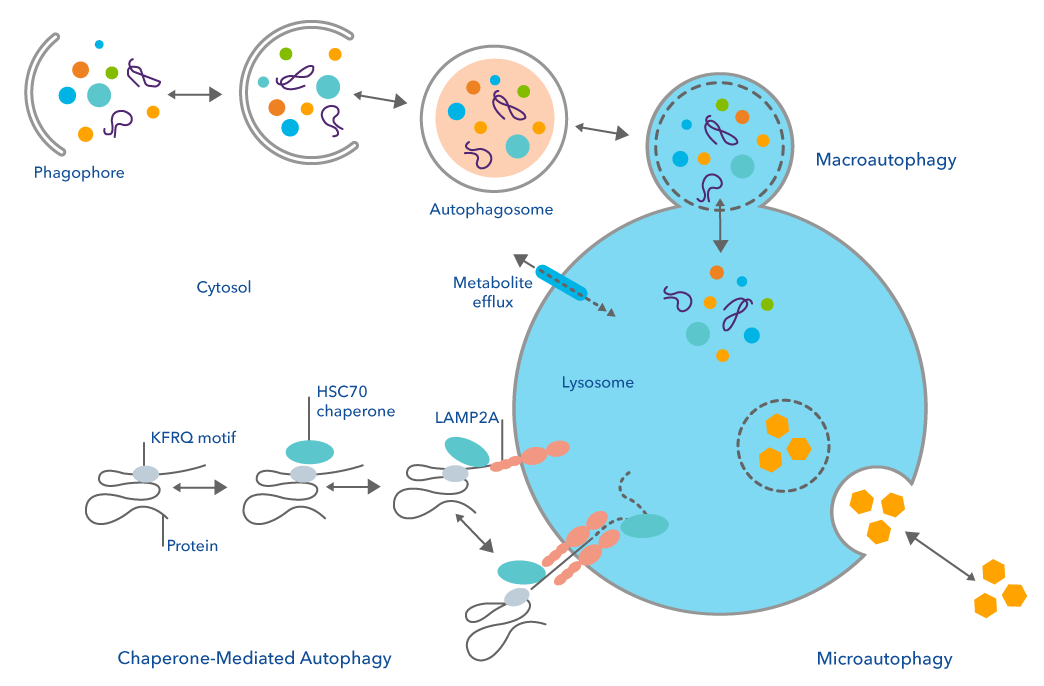
While all the forms of protein degradation and autophagy are important in disease, CMA in particular plays a diverse role in both normal biology, as well as cancer and neurodegenerative pathologies. Aberrant activation of CMA has been linked to the survival and proliferation of cancer cells. The mechanism of this regulation is tightly linked to cell metabolism, and studies have shown that inhibition of CMA restricts many rate limiting glycolytic enzymes resulting in reduced cell glycolysis and ATP production2. Additionally, it is thought that upregulated CMA may help to clear the toxic buildup of misfolded proteins during the oncogenic process allowing the cells to survive and continue to proliferate.
Alternatively, in neurodegenerative diseases, CMA plays a protective role, where it helps to facilitate the removal of deleterious proteins that are the staple pathology in many of these diseases3. CMA also plays an interesting role in ageing, one that is somewhat unique compared to other protein degradation mechanisms. A decrease in optimal CMA is associated with ageing, and has been observed in aged cells and tissues of rodents and patients4. This phenotype is directly linked to the decreased stability of LAMP-2A on the lysosomal membrane in aged cells and the subsequent retardation of protein translocation into the lysosomal lumen.
Thus far, many studies across tissue type, process, and disease state have implicated CMA in a pathology, but there is still much to be learned about the mechanisms and implications of this process across biology.
Christina Towers, PhD
-
Cuervo, A. M. & Wong, E. (2014) Chaperone-mediated autophagy: roles in disease and aging Cell Res 24:92-104.
-
Kon, M. et al. (2011) Chaperone-mediated autophagy is required for tumor growth. Sci Transl Med 3:109-117.
-
Towers, C. G. & Thorburn, A. (2016) Therapeutic Targeting of Autophagy EBioMedicine 14:15-23.
-
Cuervo, A. M. & Dice, J. F. (2000) Age-related decline in chaperone-mediated autophagy J Biol Chem 275:31505-31513.