Stem Cell Signaling
Self-renewal and differentiation potential, two key defining stem cell properties, are underscored by a network of transcription factors, epigenetic modifiers and modulating kinase signaling pathways. The Yamanaka factors, which include octamer-binding transcription factor-4 (Oct4), sex-determining region Y-box 2 (Sox2), Kruppel-like factor-4 (Klf4), and c-myelocytomatosis (c-Myc), were shown by Takahashi and Yamanaka to play a key role in the maintenance of pluripotency. Their work described for the first time the generation of induced pluripotent stem cells (iPSCs) by exogenous expression of a transcriptional pluripotency program in adult differentiated cells. Oct4, Sox2 and Nanog are now commonly considered core pluripotency factors.
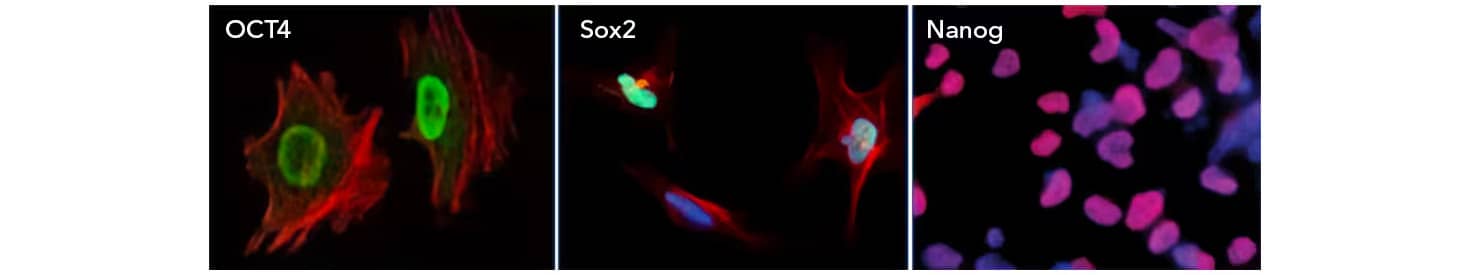
Immunocytochemistry/Immunofluorescence: (Left) OCT4 Antibody [NBP1-51664] OCT4 staining in NTERA-2 cells (Green) and actin staining with Alexa Fluor-555 phalloidin (red). (Middle) SOX2 Antibody [NB110-37235] SOX2 staining in NTERA-2 cells with Dylight 488 (green). α-tubulin and nuclei were counterstained with Dylight 550 (red) and DAPI (blue) [NBP2-31156], respectively. (Right) Nanog staining in immersion fixed BG01V human embryonic stem cells with Goat Anti-Human Nanog Antigen Affinity-purified Polyclonal Antibody (AF1997) detected with the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (NL001)(red) and counterstained with DAPI (blue).
How Core Pluripotency Factors Work?
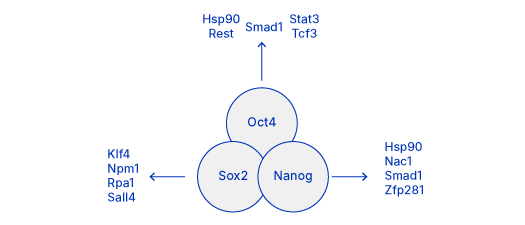
Core pluripotency transcription factors interact with each other and with many other transcriptional regulators to maintain stem cell pluripotency. These core factors engage in complex interactions to regulate their own expression and promote the expression of genes which support pluripotency or inhibit those which lead to differentiation.
Kinase Modulating Pathways
Several kinase signal transduction pathways modulate the core pluripotency transcription factors in response to both intrinsic and extrinsic stimuli, preserving stem cell self-renewal and differentiation potential. Synergy between these pathways ensures the maintenance of pluripotency.
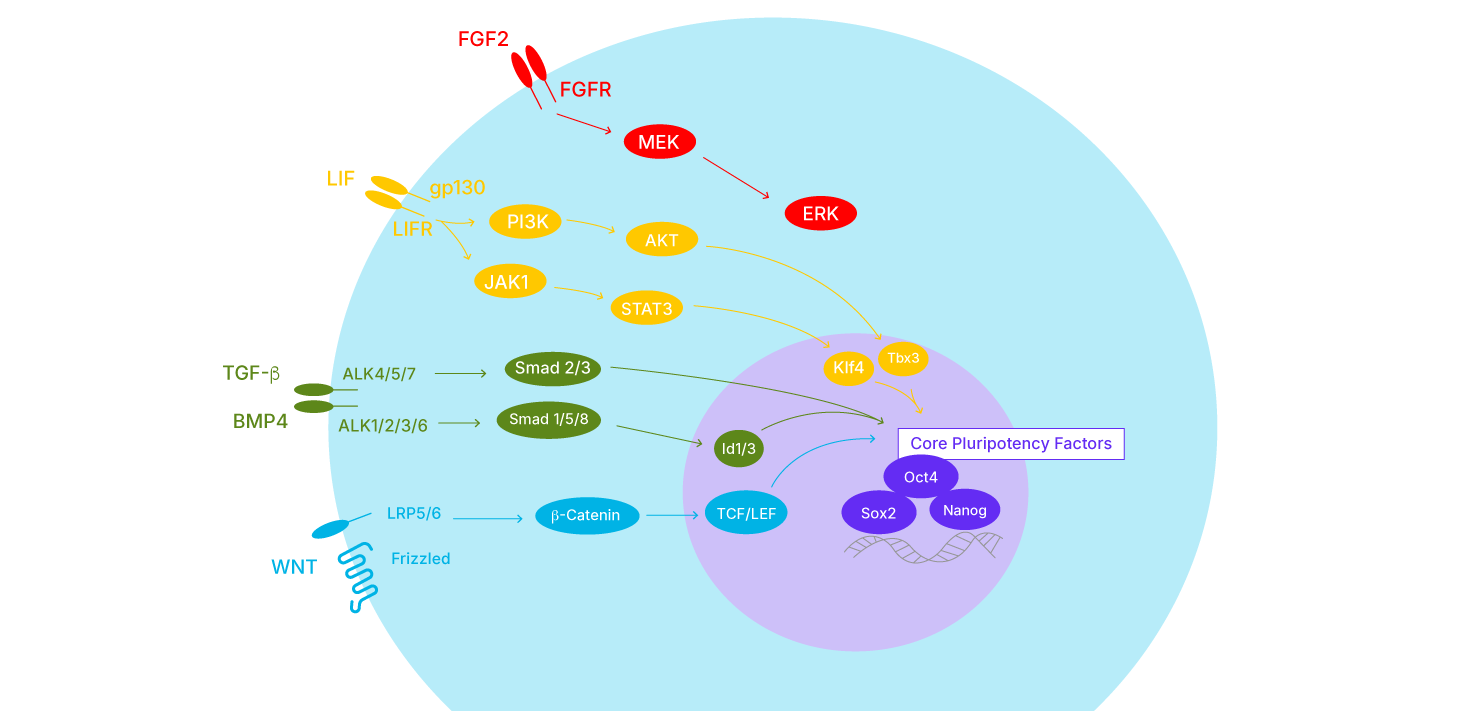
WNT signaling- Involved in the regulation of cellular proliferation, adhesion, and migration. Canonical WNT signaling leads to the stabilization of the transcriptional co-activator beta-catenin. Formation of a nuclear complex between b-catenin and T-cell factor/lymphoid enhancer factor (TCF/LEF) supports the expression of Oct4 and Nanog in pluripotent stem cells (mESC and hESC).
TGF-β signaling- Involved in the regulation of a wide range of cellular processes such as differentiation, proliferation, and programmed cell death. Important for the maintenance of self-renewal and differentiation potential of mESCs and hESCs. For example, canonical TGF-β signaling initiated by BMP4 leads to Smad1/5/8 activation and induction of Inhibitor of DNA-binding/differentiation (Id) proteins for maintenance of mESCs. In contrast, TGF-β signaling initiated by TGF-β and Activin activate Smad2 for maintenance of primed-mESCs and hESCs.
LIF signaling- The simultaneous activation of the JAK/STAT3 and PI 3-K/AKT pathways plays an important role in the maintenance of mESC pluripotency. Activation of JAK/STAT3 leads to the induction of Klf4 and expression of SOX2, while activation of the P I3-K/AKT pathway increases the expression of Nanog by induction of Tbx3.
FGF signaling- Regulates various cellular processes including cell proliferation, migration, differentiation, and survival. Activation of ERK/MAPK signaling downstream of tyrosine kinase receptor/FGF2 stimulation is involved in the maintenance of hESC pluripotency. FGF/ERK signaling is not required for mESC maintenance, rather ERK activation leads to the differentiation of mESCs.
ExCellerate™ iPSC Expansion Medium
Supports robust expansion and maintenance of pluripotent stem cell culture for enhanced consistency and reproducibility.
- Animal component-free
- No growth factor supplementation required
- Stable cell integrity over long term culture
Bieberich, E., & Wang, G. (2013) Molecular mechanisms underlying pluripotency. In Pluripotent Stem Cells. https://doi.org/10.5772/55596
Fernandez-Alonso, R., Bustos, F., Williams, C. A. C., & Findlay, G. M. (2017) Protein kinases in pluripotency—beyond the usual suspects. Journal of Molecular Biology. https://doi.org/10.1016/j.jmb.2017.04.013
Katoh, M., & Katoh, M. (2007) WNT signaling pathway and stem cell signaling network. Clinical Cancer Research. https://doi.org/10.1158/1078-0432.CCR-06-2316
Lanner, F., & Rossant, J. (2010) The role of FGF/Erk signaling in pluripotent cells. Development. https://doi.org/10.1242/dev.050146
Lee, J., Park, Y. J., & Jung, H. (2019) Protein kinases and their inhibitors in pluripotent stem cell fate regulation. Stem Cells International. https://doi.org/10.1155/2019/1569740
Mullen, A. C., & Wrana, J. L. (2017) TGF-β family signaling in embryonic and somatic stem-cell renewal and differentiation. Cold Spring Harbor Perspectives in Biology. https://doi.org/10.1101/cshperspect.a022186
Takahashi, K., & Yamanaka, S. (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. https://doi.org/10.1016/j.cell.2006.07.024
Takahashi, K., Tanabe, K., Ohnuki, M. et al. (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. https://doi.org/10.1016/j.cell.2007.11.019
Tang, Y., & Tian, X. (Cindy). (2013) JAK-STAT3 and somatic cell reprogramming. JAK-STAT. https://doi.org/10.4161/jkst.24935
Zakrzewski, W., Dobrzyński, M., Szymonowicz, M., & Rybak, Z. (2019). Stem cells: Past, present, and future. Stem Cell Research and Therapy. https://doi.org/10.1186/s13287-019-1165-5

