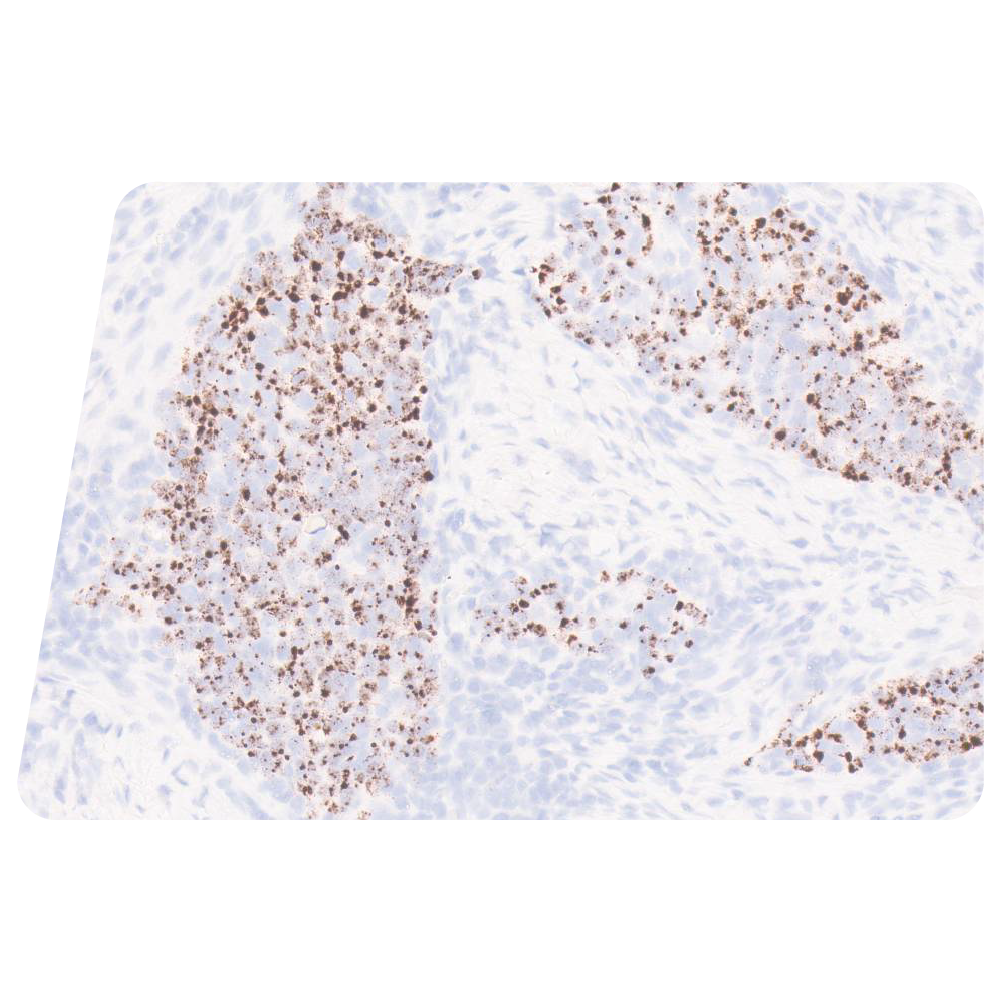
Human Papillomavirus (HPV) and Oropharyngeal Squamous Cell Carcinomas (OPSCC)
Human papillomavirus (HPV) is a major cause of oropharyngeal squamous cell carcinomas, and HPV status has emerged as a prognostic marker that significantly impacts clinical management.1 As HPV-driven carcinomas are dependent on the permanent over-expression of the HPV E6 and E7 viral oncogene mRNAs, the presence of E6/E7 mRNA is considered to be the best biomarker for identifying HPV-related head and neck cancers.2,3 Common techniques for HPV detection in OPSCC, including DNA ISH and p16 immunohistochemistry (IHC), often lead to false positives or false negatives. Previous studies reported 5–20% false positive rate of p16 IHC positivity in HPV-negative OPSCC patients.4
RNAscope in situ hybridization (ISH) gives anatomic pathologists a direct method for detection of active HPV through interrogation of E6/E7 viral oncogene mRNA, targets that provide unequivocal evidence of HPV in contrast to p16, a surrogate marker, which is upregulated by HPV as well as other non-HPV pathways. (FIGURE 1)
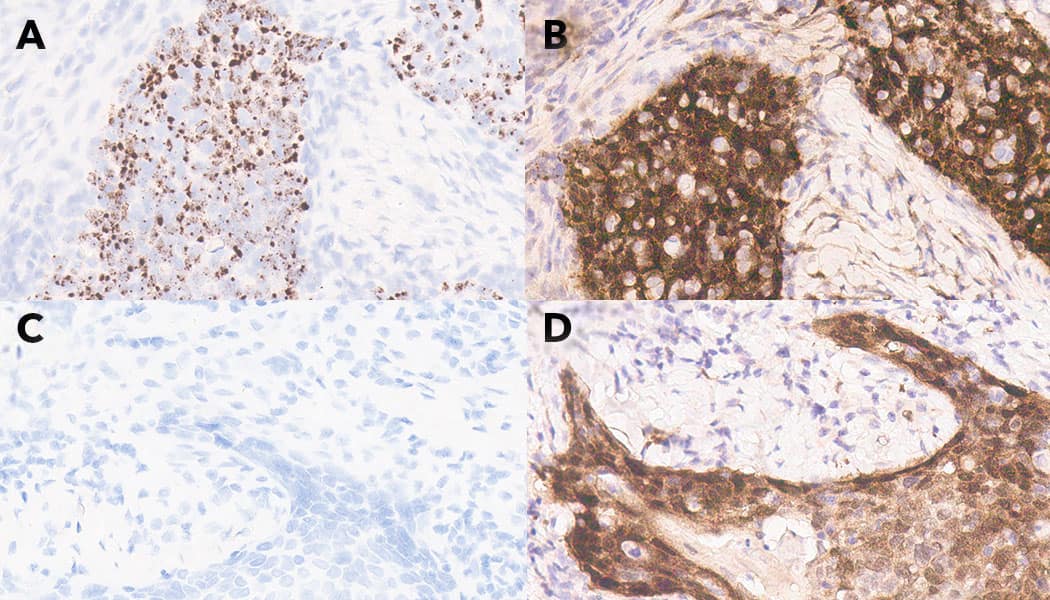
FIGURE 1: Squamous cell carcinoma (SCC) stained with RNAscope ISH Probe High Risk HPV (A and C) and p16 IHC (B and D). A and B: Tonsil SCC positive for high risk HPV mRNA by RNAscope (A) and positive for p16 by IHC (B). C and D: Tonsil SCC negative for high risk HPV mRNA by RNAscope (C) and positive for p16 by IHC (D).
The RNAscope ISH Breakthrough: When Clarity Matters
A Leica-ACD Sponsored Webinar, hosted by Pathology News
In this roundtable discussion, pathologists Dr. Monroe and Dr. Laser provide an overview of RNAscope™ technology and how it is shaping the molecular in situ hybridization (ISH) testing landscape.
- Understand the significance of ISH and its traditional role in diagnostic testing as a valuable tool for obtaining a comprehensive diagnostic picture.
- Learn about the evolution of ISH, focusing on how RNAscope technology has successfully progressed the capabilities of traditional ISH through its potential to provide higher sensitivity and specificity in RNA detection.
- See the ISH difference for yourself as RNAscope is put into context and comes alive with slide images. You will see how the technology allows for highly specific target recognition and amplification of RNA signals.
Watch Video (right) to learn more.
Detection of High-Risk HPV in FFPE Tissue Specimens Using RNAscope
The ease of adoption of RNA ISH technology on the automated Leica Biosystems BOND-III stainer (Figure 2A) has enabled laboratories to quickly establish this technology, minimizing inter-user variation and optimizing sample pretreatment. RNAscope ISH Probe High Risk HPV (Figure 2B) is for use in clinical laboratories with the CE marked BOND RNAscope Brown Detection (Figure 2C) on the BOND-III stainer. The assay qualitatively detects E6/E7 mRNA of high-risk HPV type 16, 18, 26, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 73, and 82 in FFPE tissue specimens. (Figure 3)
FIGURE 2: Automated RNAscope ISH fits seamlessly into pathology workflows.
A. Leica BOND-III fully automated IHC/ISH stainer; B. RNAscope ISH Probe High Risk HPV; C. BOND RNAscope Brown Detection
FIGURE 3: RNAscope ISH Probe High Risk HPV (cocktail including HPV types 16, 18, 26, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 73, & 82).
Tissue: Oropharyngeal squamous cell carcinoma (OPSCC).
Table 1: Comparison of RNAscope ISH Probe High Risk HPV and p16 IHC
| p16 IHC | |||
|---|---|---|---|
| Positive | Negative | ||
|
RNAscope ISH Probe High Risk HPV |
Positive | 24 | 0 |
| Negative | 3 | 20 | |
In our study, RNAscope ISH Probe High Risk HPV was compared to p16 IHC using 48 cases of oropharyngeal carcinoma. Specifically, serial sections from cases of tonsil, oropharynx, soft palate, back of the pharynx, and base of the tongue were stained with RNAscope ISH Probe High Risk HPV and control probes, as well as p16 IHC. One sample was excluded due to unacceptable RNA integrity as indicated by the UBC control probe. Comparative RNAscope and p16 results are summarized in TABLE 1. The agreement observed between RNAscope ISH Probe High Risk HPV and p16 IHC is consistent with the previously reported improved specificity for mRNA ISH.4,5
Product Ordering Information
| Product Number | Product Description | Regulatory Status | Size/Tests |
|---|---|---|---|
| 200450† | RNAscope ISH Probe High Risk HPV | CE-IVD | 14 ml/30 tests |
| 200460† | RNAscope ISH Probe UBC (Positive Control) | CE-IVD | 14 ml/30 tests |
| 200470† | RNAscope ISH Probe dapB (Negative Control) | CE-IVD | 14 ml/30 tests |
| DS9815‡ | BOND RNAscope Brown Detection | CE-IVD | 60 tests |
Product Documents
References
- Lewis JS Jr, Beadle B, et al. Human Papillomavirus Testing in Head and Neck Carcinomas: Guideline from the College of American Pathologists. Arch Pathol Lab Med. 2018 May;142(5):559-597.
- Schache, A.G., Liloglou, T., et al. (2011). Evaluation of human papilloma virus diagnostic testing in oropharyngeal squamous cell carcinoma: sensitivity, specificity, and prognostic discrimination. Clin. Cancer Res. 17, 6262–6271.
- Shi, W., Kato, H., et al. (2009). Comparative prognostic value of HPV16 E6 mRNA compared with in situ hybridization for human oropharyngeal squamous carcinoma. J. Clin. Oncol. 27, 6213–6221.
- Huanhuan W, Yuyu Z, Wei B, et al. Feasibility of immunohistochemical p16 staining in the diagnosis of human papillomavirus infection in patients with squamous cell carcinoma of the head and neck: a systematic review and meta-analysis. Front Oncol. 2020;10:524928.
- Bishop JA, Ma XJ, et al. (2012 Dec.) Detection of transcriptionally active high-risk HPV in patients with head and neck squamous cell carcinoma as visualized by a novel E6/E7 mRNA in situ hybridization method. Am J Surg Pathol. 36(12):1874-82.